2.1.1 Absolute vs. relative efficacy. In Pfizer/BioNTech’s first reported clinical trial, 43,548 participants underwent randomization, of whom 43,448 received injections. The experimental vaccine (BNT162b2) was administered to 21,720 persons, and 21,728 re- ceived placebo. Across both groups, a total of 170 COVID-19 “cases” was recorded, of which 162 occurred in the placebo group, whereas 8 cases were observed in the BNT162b2 group. Based on these figures—8/162 ≈ 5%—Pfizer proceeded to claim 95% effi- cacy. Clearly, however, this efficacy is only a relative value—in absolute terms, less than 1% of the placebo group developed COVID-19, and therefore less than 1% of the vaccine group was protected from it.
The situation is similar with the subsequent, smaller test carried out on 12-15 years old adolescents [31]. Here, the vaccine group comprised 1131 individuals, whereas the placebo group included 1129 persons. In the latter group, 16 individuals were subse- quently diagnosed with COVID-19, whereas no such cases occurred in the vaccine group. True to form, Pfizer/BioNTech converted this absolute efficacy of 1.4% to a relative one of 100%; only the latter value is highlighted in the abstract of the published study.
2.1.2 Negative impact of BNT162b2 on overall morbidity in adolescents. In the cited vaccine study on adolescents, a “case” of COVID-19 was determined as follows:
The definition of confirmed COVID-19 included the presence of ≥ 1 symptom (i.e., fever, new or increased cough, new or increased shortness of breath, chills, new or increased muscle pain, new loss of taste or smell, sore throat, diarrhea, vomiting) and being SARS-CoV-2 NAAT-positive [= PCR-positive] dur- ing, or within 4 days before or after, the symptomatic period (either at the central laboratory or at a local testing facility and using an acceptable test).
Thus, a single symptom from a laundry list of non-characteristic symptoms, plus a positive finding from an unreliable laboratory test (cf. Section 1.2.6), was deemed suffi- cient to establish the diagnosis. While the study goes on to list several clinical criteria of severe disease, it gives no indication that any test persons actually suffered any of those. It can therefore be assumed that very few non-severe, and no clinically severe cases of COVID-19 occurred in the entire test population.
In stark contrast to these numbers pertaining to the disease from which the vaccina- tion is supposed to protect, side effects from the vaccination were exceedingly common. Apart from injection site pain occurring in a high percentage of the vaccine group (79% to 86%), fatigue (60% to 66%) and headache (55% to 65%) abounded. Severe fatigue and headache were reported by several percent of the test persons. Severe headache, in par- ticular, may be associated with underlying thrombotic events (see Section 3.1.3.2). It is therefore clear that, if we consider both COVID-19 and vaccine adverse effects, overall morbidity was far greater in the vaccinated than in the placebo group.
2.1.3 Unlikely claims and contradictions in Pfizer’s evidence on efficacy. We saw above that the reported efficacy of Pfizer’s vaccine is very modest when expressed in absolute terms. Even this low efficacy, however, cannot be accepted at face value. This is apparent from the assessment reports prepared by the FDA [29] and the EMA [30].
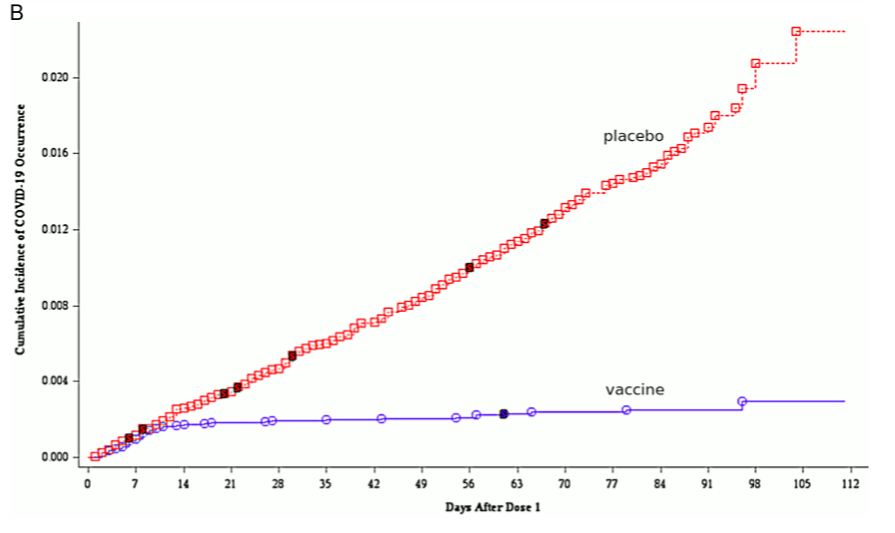
2.1.3.1 Sudden onset of immunity on day 12 after the first injection. A key illustration that occurs in both reports compares the cumulative incidence of COVID-19 among the vaccinated and the placebo group. This graph, which is shown as Figure 9 in the EMA report, is here reproduced in Figure 1B. Up to day 12 after the first injection, the cumulative incidences in the two groups track each other closely. After day 12, however, only the placebo group continues to accumulate further new cases at a steady pace, whereas the slope of the graph drops to almost zero in the vaccine group.
This remarkable observation suggests that immunity sets in very suddenly and uni- formly on day 12 exactly among the vaccinated. Since the second injection occurred 19 or more days after the first one, this would imply that one injection is enough to estab- lish full immunity. This conclusion, however, is not stated, and in fact Pfizer does not report any data at all on test persons who received one injection only.
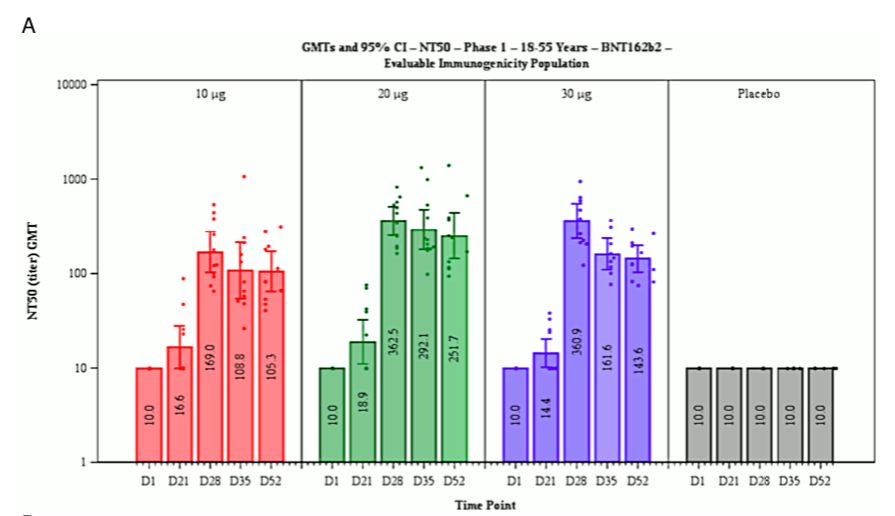
A sudden onset of full immunity on day 12 after the first exposure to the antigen is not at all a biologically plausible outcome. Typically, immunity develops more slowly and gradually; and such a pattern is in fact reported for this very same vaccine (BNT162b2) in Figure 7 of the EMA report, reproduced here as Figure 1A. The figure shows the increase of neutralizing antibodies to SARS-CoV-2 as a function of time after the first injection of the vaccine.
Figure 1 Reproduction of Figure 7 (A; neutralizing antibody titres on various days after the first injection) and of Figure 9 (B; cumulative incidence of COVID-19 among vaccinated and placebo groups) from the EMA assessment report [30]. Note the logarithmic y axis in B. See text for discussion.
Table 1 Subjects without evidence of infection in vaccine and placebo groups at various time points in the clinical trial. Data excerpted from Table 4 in [30]. See text for discussion.
The induction of neutralizing antibodies is the declared purpose of the Pfizer vaccine. Generally speaking, antibodies are protein molecules produced by our immune system when it encounters antigens—macromolecules that do not occur within our own bodies. These antigens are often part of infectious microbes, including viruses. An antibody binds to a specific feature on the surface of its antigen; this feature is called the epitope of the antibody in question.
In the context of virus infections, antibodies can be neutralizing or non-neutralizing. A neutralizing antibody recognizes an epitope that is essential for the function of the virus, for example because this epitope must make contact to a receptor molecule on the surface of the host cell which the virus must enter in order to replicate. A non- neutralizing antibody simply happens to recognize a surface feature (epitope) that plays no essential role in the infectiousness of the virus.
Considering the foregoing, we should expect that the blood level of neutralizing antibodies should reflect the degree of clinical immunity to the virus. This is, however, not at all what we see in Figure 1A. On day 21 after the first injection, that is, a full 9 days after the purported sudden onset of full clinical immunity, the amount of neutralizing antibodies in the blood has barely risen above the background level. The maximal level of neutralizing antibodies is observed only on day 28 after the first injection, at which time most test persons would already have had their second injection. The time course of cellular (T-cell) immunity was not reported, but in the absence of proof positive to the opposite it can be assumed to resemble that of the antibody response.
It is very difficult to reconcile the two contrasting observations of sudden onset of full clinical immunity on day 12, but neutralizing antibodies appearing only weeks later. Yet, neither the EMA reviewers nor those of the FDA appear to have been interested in the problem.
2.1.3.2 The Pfizer documentation contradicts itself on COVID-19 incidence after vaccination.
Table 1 lists the percentages of subjects in the vaccine group and the placebo group who showed no evidence of SARS-CoV-2 infection on day 0 (before the first dose) and on day 14 after the second dose, respectively. From the differences between the two time points, we can work out that 7.5% of the subjects in the vaccine group and 8% in the control group converted from negative to positive—that is, became infected—between the two time points.
According to [29], the second dose was administered approximately 21 days after the first, although all subjects who received it between days 19 and 42 after the first injection were included in the evaluation. If we take day 35 after the first injection as the approximate time point of the comparison, we see from Figure 1B that the cumulative incidence between day 0 and day 35 is more than twice higher in the placebo group than in the vaccine group; but from Table 1, we see that it is almost the same. Moreover, with both groups the numbers are substantially higher in the table than in the figure.
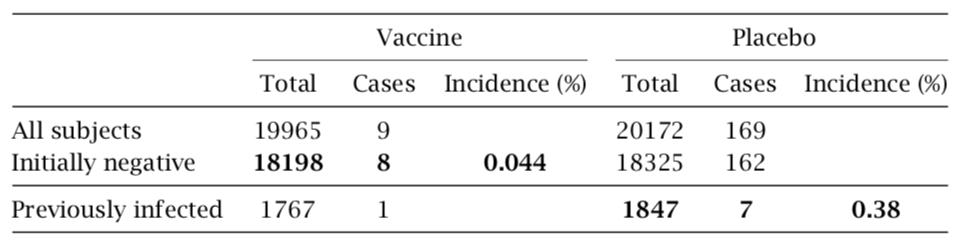
Table 2 Incidence of COVID-19 among subjects not previously infected but vaccinated, or previously infected but not vaccinated. Data excerpted from Tables 6 and 7 in [29]. See text for discussion.
These two sets of data cannot possibly be reconciled; one must be false. Since, as discussed, the sudden onset of immunity implied by Figure 1B lacks any biological plau- sibility, it is most likely that it is this data set which was fabricated.
2.1.3.3 Pfizer’s data imply that the vaccine protects from COVID more effectively than does prior infection with the virus. We can also scrutinize Pfizer’s reported data in order to compare the immunity conferred by the vaccine to that induced by prior natural infection with the virus. The relevant data are summarized in Table 2. The reported 8 cases of COVID-19 among vaccinated persons who had initially tested negative for the virus amount to an incidence of 0.044%. Pfizer also reports 7 cases among persons who had initially tested positive but were not vaccinated. Since this group is considerably smaller, those 7 cases translate into an almost ninefold higher incidence (0.38%).
It is common knowledge that vaccines will at best approach, but not surpass the im- munity conferred by the corresponding natural infection. Very robust immunity after prior natural infection with SARS-CoV-2 has recently been reported [10]; in that study, not a single case of COVID-19 was observed among 1359 individuals who had remained unvaccinated. Robust immunity after infection is also confirmed by comprehensive lab- oratory investigations [11]. Therefore, the above analysis corroborates yet again that the trial results reported by Pfizer cannot be trusted. That neither the FDA nor the EMA picked up on any of these inconsistencies does not instil confidence in the thoroughness and integrity of their review processes.
2.2 What evidence is lacking to make the case? We had already mentioned the specious and contrived character of the endpoint used in Pfizer’s clinical trials—namely, the count- ing of a COVID-19 “case” based on nothing more than a positive PCR result, together with one or more items from a list of mostly uncharacteristic clinical symptoms. We must therefore ask if the vaccine provides any benefits that are more substantial than the claimed—but, as discussed above, most likely fabricated—reduction in the count of such trivial “cases.”
2.2.1 Prevention of severe disease and mortality. Page 48 of the FDA report sums up this question as follows: “A larger number of individuals at high risk of COVID-19 and higher attack rates would be needed to confirm efficacy of the vaccine against mortality.”
We note that this quote not only answers the posed question in the negative, but it also disposes of the entire pretext for granting emergency use authorization for this experimental vaccine. If in a study that involves 40,000 individuals the number of fatal outcomes is too small to permit the detection of any benefit of the vaccine, then surely no “emergency” exists that would justify the very grave risks, and meanwhile manifest harm, associated with the extraordinarily rushed introduction of this and other COVID- 19 vaccines.
No fatalities at all occurred in the cited study on adolescents [31]; and we already noted that this study does not report any cases of severe disease either. Therefore, in this specific age group, too, neither a meaningful benefit nor an emergency are in evidence.
2.2.2 Effectiveness for those at high-risk of severe COVID-19. Here, the FDA report has this to say: “Although the proportion of participants at high risk of severe COVID- 19 is adequate for the overall evaluation of safety in the available follow-up period, the subset of certain groups such as immunocompromised individuals (e.g., those with HIV/AIDS) is too small to evaluate efficacy outcomes.”
The report shirks the question of risk reduction among those with more common predisposing conditions, such as for example chronic heart or lung disease. Naturally, the clinical study on adolescents [31] is completely barren in this regard. Overall, no evidence has been adduced by Pfizer’s clinical studies to prove clinical benefit in those at high risk of severe COVID-19.
2.2.3 Effectiveness against long-term effects of COVID-19 disease. The FDA report’s verdict is as follows: “Additional evaluations will be needed to assess the effect of the vaccine in preventing long-term effects of COVID-19, including data from clinical trials and from the vaccine’s use post authorization.” In other words, the clinical trials pro- vided no such evidence.
2.2.4 Reduction of transmission. On this topic, the FDA report offers only that “addi- tional evaluations including data from clinical trials and from vaccine use post-autho- rization will be needed to assess the effect of the vaccine in preventing virus shedding and transmission, in particular in individuals with asymptomatic infection.”
In plain language, there is no evidence that transmission is reduced, and in fact the trials were simply not even designed to prove or disprove such an effect.
2.2.5 Duration of protection. The FDA report correctly states (on page 46) that “as the interim and final analyses have a limited length of follow-up, it is not possible to assess sustained efficacy over a period longer than 2 months.” Even if we choose to believe that any efficacy at all has been demonstrated pertaining to the two-month study period, such a short duration of protection does not justify the risks associated with vaccination.
2.2.6 Inadequate efforts to determine the optimal dose. Figure 1A shows that the level of neutralizing antibodies is virtually the same with vaccine (mRNA) doses of 20μg and 30μg, respectively. This raises the question why the higher dose was employed throughout—and not only with adults, on whom these data were obtained, but also with children, whose lower body weights should suggest a dose reduction. Furthermore, the data in Figure 1B suggest that full immunity is induced already by the first dose; appli- cation of the second dose does not change the pace at which new cases accrue in the vaccine group, and therefore apparently has no effect on immunity. This would imply that a one-dose regimen should have been evaluated, which would reduce the overall likelihood of adverse events.
2.2.7 Summary. The clinical trials carried out by Pfizer contain no proof of any benefit conferred by the vaccine with respect to any clinically relevant endpoints. This applies to all tested age groups, and in particular also to adolescents.
3 The Pfizer COVID-19 vaccine lacks safety
3.1 What does the evidence show? The clinical trials for Comirnaty (BNT162b2), as well as for the other COVID-19 vaccines, were rushed through in a very short time; this has meant that proper precautions to ensure their safety were not taken. However, animal experiments carried out before the start of clinical testing already gave reason to expect severe toxicity. Unfortunately, this expectation has been abundantly borne out in practice since the beginning of mass vaccinations.
3.1.1 Preclinical data from animal experiments indicate potential for grave harm.
Comirnaty, like all other gene-based COVID-19 vaccines, causes the expression in vivo of one specific protein of SARS-CoV-2—namely, the so-called spike protein, which is lo- cated on the surface of the virus particle. The spike protein mediates the virus particle’s initial attachment to the host cell and also its subsequent entry into the cell. The key idea behind the Comirnaty vaccine is as follows:
- a synthetic mRNA that encodes the spike protein is complexed with a mixture of neutral and cationic (positively charged) synthetic lipids, which cluster together in lipid nanoparticles (LNPs);
- after injection, the LNPs facilitate the uptake of the mRNA into host cells, where the mRNA will cause the expression (synthesis) of the spike protein;
- the spike protein will appear on the surface of the host cells and induce an immune reaction to itself.The immune reaction to the spike protein will comprise both antibodies, which may or may not be neutralizing (see Section 2.1.3.1), and T-lymphocytes (T-cells). Some of these T-cells are cytotoxic (also known as T-killer cells); their function is to kill virus- infected body cells.
While this vaccination strategy may look good on paper, it has a number of drawbacks and risks. These arise both from the lipid mixture and from the spike protein, both of which have known toxic activities.
3.1.1.1 Toxic and procoagulant activities of the spike protein. Severe clinical COVID- 19 disease is often accompanied by a pathological activation of blood clotting [32]. The central role of the spike protein in this complication is recognized [33]. Notably, there are at least two different mechanisms for triggering blood coagulation:
- If the spike protein is expressed within vascular endothelial cells—the innermost cell layer of the blood vessels—then an immune reaction to the spike protein can destroy these cells. The resulting vascular lesion will activate blood clotting. This immune reaction can involve cytotoxic T-cells, but also antibodies that trigger the complement system and other immune effector mechanisms.
- Spike protein molecules that are formed within the circulation, or which enter it after being synthesized elsewhere in the body, can directly bind to blood platelets (thromboycytes) and activate them. This will again set off blood clotting.
The second mechanism is significant because it does not involve an immune reaction; therefore, it can be triggered right away even in those persons who have no pre-existing immunity. The first mechanism will be most effective in those who already have immunity to the spike protein, due to either infection with the virus or a previous injection of vaccine. Note that the underlying mechanism of cell damage will also operate in other tissues—any cell in the body that expresses the spike protein will thereby become a tar- get for the immune system.
Since Comirnaty and other gene-based vaccines induce the synthesis of active, and therefore potentially toxic, spike protein, it is important to understand how this protein with be distributed within the body. Toxicity might be limited if the vaccine, and there- fore the synthesis of the spike protein, remained confined to the site of injection, within the muscle tissue but outside the circulation. On the other hand, if the vaccine were to enter the bloodstream, then one would have to expect expression of the spike protein within the blood vessels and toxicity through the activation of blood clotting.
3.1.1.2 Distribution of the vaccine in animal experiments. As it turns out, the vac- cine does indeed appear in the bloodstream very rapidly after intramuscular injection. In experiments which Pfizer reported to the Japanese health authorities [34], rats were injected with a mock vaccine sample. This material was was chemically similar to Comir- naty, but it contained an mRNA molecule that encoded an easily traceable, non-toxic model protein (luciferase) rather than the SARS-CoV-2 spike protein. The lipid mixture used to form the LNPs was the exact same as with Comirnaty. One of the lipids in this mixture was radioactively labelled, which permitted the distribution of the sample within the body to be traced and quantified sensitively and accurately. Several remarkable ob- servations were made:
- The radioactive lipid appeared rapidly in the bloodstream. The blood plasma concen- tration peaked after 2 hours; but even at only 15 minutes into the experiment, the plasma level had already reached 45% of that maximal value.
- Very high levels of the radioactive lipid accumulated in the liver, the spleen, the adrenal glands, and the ovaries.
- Comparatively low levels accumulated in the central nervous system (the brain and the spinal cord).
- Expression of the model protein encoded by the mRNA was studied only in the liver, where it was readily detected.
3.1.1.3 Mechanism of vaccine uptake into the bloodstream. Considering that the com- plex consisting of mRNA with bound LNPs has a rather large molecular size, we must ask how it managed to enter the bloodstream so rapidly. After intramuscular injection, the bulk of the vaccine should end up in the “interstitial” space, that is, the extracellular space outside the blood vessels. This space is separated from the intravascular space (the circulation) by the capillary barrier, which permits free passage only to small mo- lecules such as oxygen or glucose (blood sugar) but is impermeable to large molecules such as plasma proteins; and the vaccine particles would be even larger than those.
The fluid within the interstitial space is continuously drained through the lymphatic system; all lymph fluid ultimately enters the bloodstream through the thoracic duct. Par- ticles which are too large for traversing the capillary barrier can ultimately reach the circulation by way of this lymphatic drainage. However, this process tends to be consid- erably slower [35] than was observed here with the model vaccine. We must therefore ask if the model vaccine may have broken down the capillary barrier and thereby gained direct entry to the bloodstream.
Lipid mixtures similar to those contained in the Pfizer vaccine have been used exper- imentally to penetrate the blood brain barrier after intravenous injection [36]. The blood brain barrier can be described as a “fortified version” of the regular capillary barrier—if it can be broken down, then we must expect the same with a regular capillary barrier, too. The high local concentration of the lipid nanoparticles that will result after intra- muscular injection will further promote the breakdown of the barrier. The upshot of this is that the vaccine will appear in the bloodstream, in large amounts and on short order. Complications due to blood clotting must therefore be expected.
3.1.1.4 Other indications of LNP toxicity. The proposed breakdown of the capillary barrier by the LNPs implies a cytotoxic effect on the endothelial cells, which form the only cellular element of the capillary walls. Cytotoxic effects of the LNPs are also evident from damage to muscle fibres at the injection site [30, p. 49] and to liver cells [30, p. 46]. Note that these data, too, were obtained with the model mRNA encoding the presumably non-toxic luciferase enzyme. Therefore, these cytotoxic actions are not due to any direct action of the spike protein. An immunological component of the cell damage cannot be completely ruled out, but it is likely not dominant in this case, since luciferase, unlike spike protein, is not transported to the cell surface.
3.1.1.5 Mechanisms of accumulation in specific organs. The high rates of accumulation of the vaccine in the liver and the spleen suggest uptake by macrophage cells, which abound in both organs and are generally in charge of clearing away unwanted de- bris. The accumulation in the adrenal glands, the ovaries, and again the liver suggests a role of lipoproteins in cellular uptake within these organs. Lipoproteins are complexes of lipids and specific protein molecules (apolipoproteins) that function as lipid carriers in the bloodstream. The liver has a central role in lipid and lipoprotein metabolism generally, whereas the adrenal glands and the ovaries take up lipoproteins to acquire cholesterol, which they then convert to their respective steroid hormones. Such a role of lipoproteins in the transport and cellular uptake of lipid nanoparticles is in fact accepted [37]. We must therefore expect that other organs with a high rate of lipoprotein uptake will be similarly affected. This includes in particular the placenta, which like the ovaries produces large amounts of steroid hormone (progesterone), and the lactating mammary glands, which acquire cholesterol contained in lipoproteins for secretion into the breast milk.
3.1.1.6 Correlation of lipid uptake and mRNA expression. In the experimental study in question, the liver was also shown to express the mRNA that is associated with the LNPs (see [30], Section 2.3.2). As stated above, the mRNA used in this study encoded the firefly enzyme luciferase, which is the very protein that enables these animals to glow in the dark. Mammalian tissues expressing this enzyme will also become luminescent, in proportion to the amount of luciferase protein which they synthesize. Measurements of this luminescence are not very sensitive, though, which was most likely the reason why Pfizer carried them out only with the liver but not with other, smaller organs. However, in the absence of proof positive to the opposite, we must assume that the correlation between efficient LNP uptake and mRNA expression that applies to the liver will also hold with other organs. If the cargo mRNA encodes the spike protein, then these organs will be exposed to the toxicity of the spike protein, and to the immune reaction against it, in proportion to the level of LNP and mRNA uptake.
3.1.1.7 Potential risks to fertility and to the breastfed newborn. A high level of expression of spike in the ovaries raises the prospect of significant damage to that organ, with possible consequences for female fertility. Uptake of the vaccine by mammary gland cells opens two possible pathways of toxicity to the breastfed child: firstly, the expression of spike protein and its secretion into the breast milk, and secondly, the wholesale transfer of the vaccine into the milk. The mammary glands are apocrine, which means that they pinch off and release fragments of their own cytoplasm into the milk; thus, anything that has reached the cytoplasm might also reach the breast milk. In this connection, we note that both the VAERS database and the EU drug adverse events registry (EudraVigilance) report fatalities in breastfed newborns after vaccination of their mothers (see Section 3.1.3.6).
3.1.1.8 Pfizer’s failure to investigate risks evident from preclinical investigations.
With the exception of fertility, which can simply not be evaluated within the short period of time for which the vaccines have been in use, all of the risks discussed above have been substantiated since the vaccines have been rolled out—all are manifest in the re- ports to the various adverse event registries (see Section 3.1.3). We must stress again that each of these risks could readily be inferred from the cited limited preclinical data, but were not followed up with appropriate in-depth investigations. In particular, the clinical trials did not monitor any laboratory parameters that could have provided information on these risks, such as those related to blood coagulation (e.g. D-dimers/thrombocytes) or liver damage (e.g. γ-glutamyltransferase).
3.1.2 Contaminations arising from the manufacturing process. The commercial scale manufacturing process of BNT162b2 gives rise to several contaminations that may com- promise vaccine safety and effectiveness. For brevity, we will here mention only two such contaminants.
3.1.2.1 Contaminating bacterial DNA. The mRNA is produced in vitro using a DNA template, which in turn is obtained from bacterial cells. While steps are taken to remove this DNA afterwards, they are not completely effective, which is acknowledged in the EMA report (pages 17 and 40). Contaminating DNA injected with the vaccine may insert into the genomes of host cells and cause potentially harmful mutations. Bacterial DNA also non-specifically promotes inflammation.
3.1.2.2 Lipid impurites. The EMA report also observes impurities originating from the synthesis of the lipid ingredients of the vaccine (page 24):
Lipid-related impurities have been observed in some recently manufactured finished product batches, correlated with ALC-0315 lipid batches. The quality of ALC-0315 excipient is considered acceptable based on the available data on condition that specific impurities in the finished product will be further evaluated.
Considering that the synthetic lipid referred to as ALC-0315 has never before been used on humans, there is no sound empirical basis for deciding on “acceptable” levels of impurities. Furthermore, it appears that the contaminating species have not even been identified. EMA’s arbitrary blanket approval of unknown contaminants of an unproven vaccine ingredient is completely unacceptable.
3.1.3 Adverse events after the onset of vaccinations. Since the introduction of the vaccines, numerous adverse events have been reported to registries around the world. We will here focus on two registries, namely, the U.S. vaccine adverse events reporting system (VAERS) and the EU monitoring system for drug adverse events (EudraVigilance). All numbers quoted below are as of May 21st unless stated otherwise.
3.1.3.1 Fatalities reported in connection with COVID vaccines. Within just five months of the onset of vaccinations, EudraVigilance has accumulated 12,886 deaths in connection with the COVID-19 vaccines, of which the Pfizer vaccine accounted for almost half (6,306). In the same time period, VAERS has run up 4,406 deaths in all; of these, 91% were associated with the mRNA vaccines, with Pfizer accounting for 44% and Moderna for 47% of the total.
It is impossible to know what percentage of all fatalities that occur after vaccina- tion will actually be reported to VAERS or EudraVigilance. However, note that the 4,406 COVID vaccine-related fatalities accrued by VAERS during just the past 5 months exceed the cumulative total of all other vaccines combined, over the entire previous 20 years. It is therefore clear that these vaccines are far and away the most deadly ones in history— quite predictably so, and all for a disease whose case fatality rate does not exceed that of influenza [1, 38].
3.1.3.2 Severe events related to disrupted blood clotting. The litany of diagnoses in both databases that indicate pathological activation of blood clotting is almost endless— heart attacks, strokes, thromboses in the brain and in other organs, pulmonary em- bolism; but also thrombocytopenia and bleeding, which result from excessive consump- tion of thrombocytes and of coagulation factors in disseminated intravascular coagula- tion. These disease mechanisms caused many of the fatalities summarized above; in other cases, they caused severe acute disease, which will in many cases leave behind severe disability.
3.1.3.3 Other severe reactions. Severe reactions also include seizures, other neurolog- ical symptoms, particularly related to motor control, and severe systemic inflammation with damage to multiple organs. Again, in many of these patients, long-lasting or even permanent residual damage is highly likely.
3.1.3.4 Severe adverse reactions among adolescents. In the age group of 12-17 years, two deaths likely related to the Pfizer vaccine were already reported to EudraVigilance. Also in this age group, there were 16 cases of myocarditis, all in males, and 28 cases of seizures among both sexes, 3 of them reported as life-threatening. There also were a few cases of stroke, myocardial infarction, and severe inflammatory disease.
While the numbers of adverse events are much lower than those among adults, this is simply due to the hitherto far lower rates of vaccination in this age group. Should systematic vaccination be green-lighted for adolescents, we must expect these numbers to rapidly climb to a level resembling that seen in adults.
3.1.3.5 Miscarriages. As of June 21st, 2021, EudraVigilance lists 325 cases of miscar- riage among vaccinated pregnant women. While it is difficult to ascertain by just how much vaccination will raise the rate of miscarriage, most of these cases were reported by healthcare professionals, who evidently considered a connection to the vaccine at least plausible. This series of cases alone would be reason enough to pause the vaccinations and investigate.
3.1.3.6 Deaths among breastfed infants. Although it does not directly relate to the age group which is the focus of this lawsuit and this expert opinion, it bears mention that both VAERS and EudraVigilance contain reports of death among breastfed children shortly after their mothers had received the Pfizer vaccine.
In Section 3.1.1.5, we discussed the possibility of vaccine uptake into the placenta and the breast glands. The reported miscarriages and fatalities in newborns indicate that these risks must be taken very seriously, and that Pfizer acted negligently in not investigating them in any of their reported preclinical and clinical trials.
3.2 Missing evidence. We saw above that significant positive indications of risk were neglected in the clinical trials and subsequent rushed emergency approval of the Pfizer vaccine, with unfortunate yet predictable outcomes. Equally damning is the list of omissi- ons—potential risks that should have been investigated in preclinical or clinical trials but never were.
3.2.1 Proper pharmacokinetics. Section 3.1.1.2 described some experiments pertaining to the distribution of a surrogate vaccine. While these studies did provide important and useful information, it must be noted that the expression of the spike protein instead of the presumably inert luciferase enzyme might affect the distribution due to its interfer- ence with vascular integrity, including at the blood brain barrier, and with blood clotting. EMA and other regulators should have insisted that such experiments be carried out and documented.
3.2.2 Drug interactions. The EMA report states (page 110): Interaction studies with other vaccines have not been performed, which is acceptable given the need to use the vaccine in an emergency situation.
Since it is clear that mortality due to COVID-19 is low (see Section 1.1.1) and therefore that no emergency exists, this argument must be rejected as specious.
Immunosuppressive effects of BNT162b2 are apparent from a drop of blood lym- phocyte numbers among those vaccinated, as well as from clinical observations of Her- pes zoster (shingles), which arises through the reactivation of persistent varicella-zoster virus [39]. This suggests that the desired immune response to other vaccines simultane- ously administered may be impaired.
Furthermore, studies of interactions should not have been limited to vaccines alone, but also been extended to other drugs. One area of concern is the experimentally ap- parent liver toxicity of BNT162b2. The liver is central in the metabolic inactivation and disposal of many drugs; any interference with the function of this organ immediately creates numerous possibilities of adverse drug interactions.
3.2.3 Genotoxicity. No studies have been carried out regarding genotoxicity, that is, damage to the human genetic material, which could lead to heritable mutations and cancer. In the EMA report [30, p. 50], this is justified as follows:
No genotoxicity studies have been provided. This is acceptable because the components of the vaccine formulation are lipids and RNA, which are not expected to have genotoxic potential. The risk assessment performed by the ap- plicant shows that the risk of genotoxicity related to these excipients [i.e. the synthetic lipids] is very low based on literature data.
In reality, it is known that the LNPs contained in BNT162b2 can enter all kinds of cells—that is, after all, the purpose of their inclusion in this vaccine preparation. It is also known that, once inside the cell, cationic lipids disrupt mitochondrial function (cell respiration) and cause oxidative stress, which in turn leads to DNA damage.
It should be mentioned that two of the lipids used by Pfizer—namely, the cationic lipid ALC-0315 and the PEGylated lipid ALC-0159, which account for 30-50% and for 2- 6%, respectively, of the total lipid content—had not previously been approved for use in humans. Pfizer’s and EMA’s cavalier attitude to the use of novel and so far unproven chemicals as components in drug or vaccine preparations without comprehensive studies on toxicity, including genotoxcicity, is completely unscientific and unacceptable.
3.2.4 Reproductive toxicity. Reproductive toxicity was assessed using only one species (rats) and on only small numbers of animals (21 litters). A greater than twofold increase in pre-implantation loss of embryos was noted, with a rate of 9.77% in the vaccine group, compared to 4.09% in the control group. Instead of merely stating [30, p. 50] that the higher value was “within historical control data range,” the study should have stated un- ambiguously whether or not this difference was statistically significant; and if it was not, the number of experiments should have been increased to ensure the required statistical power. The same applies to the observations of “very low incidence of gastroschisis, mouth/jaw malformations, right sided aortic arch, and cervical vertebrae abnormalities.” Overall, these studies are inadequately described and apparently were also inadequately carried out.
3.2.5 Autoimmunity. Exposure to the vaccine will lead to cell damage due to the cationic lipids, and also to the immune attack on cells producing the spike protein. From the cells undergoing destruction, proteins and other macromolecules will be released; such mate- rial must then be cleared away by macrophages.
When the clearing system is overloaded because of excessive cell damage and apoptosis (cell death), then the accumulation of cellular debris will lead to chronically excessive type I interferon release; this, in turn, will trigger further inflammation. With time, some macromolecules in the debris will become targets for the formation of autoanti- bodies and the activation of autoreactive cytotoxic T cells—they will begin to function as auto-antigens. This then leads to further tissue damage and the release of more auto- antigens—autoimmune disease will develop. Such an outcome is particularly likely in im- munocompromised people or in those who are genetically predisposed to autoimmune disease (e.g. those with the HLA-B27 allele).
The risk of autoimmunity induced by BNT162b2 could be adequately addressed only in long-term studies; as with fertility or cancer, the very short period of preclinical and clinical testing means that we are flying blind. It should go without saying that all of these risks are particularly grave with children, adolescents, and young adults.
3.2.6 Antibody-dependent enhancement. While antibodies in principle serve to protect us from infections, in some cases they can increase disease severity. This phenomenon is referred to as antibody-dependent enhancement.
3.2.6.1 The principle. In Section 2.1.3.1 above, we saw that antibodies may or may not neutralize the virus that elicited them. While in most cases non-neutralizing antibodies are not harmful, with some viruses they can actually make matters worse by facilitating entry of these viruses into host cells. This occurs because certain cells of the immune system are supposed to take up antibody-tagged microbes and destroy them. If a virus particle to which antibodies have bound is taken up by such a cell but then manages to evade destruction, then it may instead start to multiply within this cell. Overall, the antibody will then have enhanced the replication of the virus. Clinically, this antibody- dependent enhancement (ADE) can cause a hyperinflammatory response (a “cytokine storm”) that will amplify the damage to our lungs, liver and other organs of our body.
ADE can occur both after natural infection and after vaccination, and it has been observed with several virus families, including Dengue virus, Ebola virus, respiratory syncytial virus (RSV), and HIV [40]. Importantly, ADE also occurs with coronaviruses, and in particular with SARS, whose causative agent is closely related to SARS-CoV-2. Attempts to develop vaccines to SARS repeatedly failed due to ADE—the vaccines did induce antibodies, but when the vaccinated animals were subsequently challenged with the virus, they became more ill than the unvaccinated controls (see e.g. [41]).
3.2.6.2 SARS-CoV-2 and ADE. The possibility of ADE in the context of natural infection with SARS-CoV-2, as well as of vaccination against it, has been acknowledged [42]. More specifically, ADE due to spike protein antibodies elicited by other coronavirus strains has been invoked to account for the peculiar geographical distribution of disease severity within China [43]. However, the experimental research required to address it remains missing, even after more than one year into the pandemic.
With some experimental SARS vaccines, ADE could be mitigated through the use of inulin-based adjuvants [44]. This approach might be feasible for avoiding ADE with COVID-19 vaccines also, but so far this appears not to have been investigated with any of the existing COVID vaccines.
Pfizer and the regulatory bodies are well aware of the risk of ADE as well. The FDA notes in its briefing document [29, p. 44]:
Pfizer submitted a Pharmacovigilance Plan (PVP) to monitor safety concerns that could be associated with Pfizer-BioNTech COVID-19 Vaccine. The Sponsor identified vaccine-associated enhanced disease including vaccine-associated enhanced respiratory disease as an important potential risk.
Here, the term “vaccine-associated enhanced disease” refers to ADE. EMA has likewise acknowledged that this risk must be investigated further [30, p. 141]:
Any important potential risks that may be specific to vaccination for COVID- 19 (e.g. vaccine associated enhanced respiratory disease) should be taken into account. The Applicant has included VAED/VAERD as an important potential risk and will further investigate it in the ongoing pivotal study and a post- authorization safety study.
Overall, it is clear that the risk of ADE is recognized in theory but is not addressed in practice. Given the abundant evidence of ADE with experimental SARS vaccines, this is completely irresponsible.
*
Note to readers: Please click the share buttons above or below. Follow us on Instagram, @crg_globalresearch. Forward this article to your email lists. Crosspost on your blog site, internet forums. etc.
Michael Palmer MD is Associate Professor in the Department of Chemistry at the University of Waterloo, Ontario, Canada. He studied Medicine and Medical Microbiology in Germany and has taught Biochemistry since 2001 in Canada. His focus is on Pharmacology, metabolism, biological membranes and computer programming, with an experimental research focus on bacterial toxins and antibiotics (Daptomycin). He has written a textbook on Biochemical Pharmacology.
Sucharit Bhakdi MD is Professor Emeritus of Medical Microbiology and Immunology and Former Chair at the Institute of Medical Microbiology and Hygiene, Johannes Gutenberg University of Mainz.
Stefan Hockertz is Professor of Toxicology and Pharmacology, a European registered Toxicologist and Specialist in Immunology and Immunotoxicology. He is CEO of tpi consult GmbH.
All three are founding signatories of Doctors for Covid Ethics
Notes
- [1] J. P. A. Ioannidis: Infection fatality rate of COVID-19 inferred from seroprevalence data. Bull. World Health Organ. (2020), BLT.20.265892. url: https://www.who.int/bulletin/ online_first/BLT.20.265892.pdf.
- [2] J. P. A. Ioannidis: Reconciling estimates of global spread and infection fatality rates of COVID-19: An overview of systematic evaluations. Eur. J. Clin. Invest. 5 (2021), e133554. pmid: 33768536.
- [3] CDC COVID-19 Response Team: Coronavirus Disease 2019 in Children – United States, February 12-April 2, 2020. MMWR. Morbidity and mortality weekly report 69 (2020), 422– 426. pmid: 32271728.
- [4] S. Tsabouri et al.: Risk Factors for Severity in Children with Coronavirus Disease 2019: A Comprehensive Literature Review. Pediatric clinics of North America 68 (2021), 321–338. pmid: 33228941.
- [5] J. Y. Abrams et al.: Multisystem Inflammatory Syndrome in Children Associated with Severe Acute Respiratory Syndrome Coronavirus 2: A Systematic Review. J. Pediatr. 226 (2020), 45– 54. pmid: 32768466.
- [6] P. A. McCullough et al.: Multifaceted highly targeted sequential multidrug treatment of early ambulatory high-risk SARS-CoV-2 infection (COVID-19). Reviews in cardiovascular medicine 21 (2020), 517–530. pmid: 33387997.
- [7] C. Bernigaud et al.: Oral ivermectin for a scabies outbreak in a long-term care facility: po- tential value in preventing COVID-19 and associated mortality. Br. J. Dermatol. 184 (2021), 1207–1209. pmid: 33454964.
- [8] Anonymous: WHO advises that ivermectin only be used to treat COVID-19 within clinical trials. 2021. url: https://www.who.int/news-room/feature-stories/detail/who- advises-that-ivermectin-only-be-used-to-treat-covid-19-within-clinical- trials.
- [9] J. Flood et al.: Paediatric multisystem inflammatory syndrome temporally associated with SARS-CoV-2 (PIMS-TS): Prospective, national surveillance, United Kingdom and Ireland, 2020. The Lancet regional health. Europe 3 (2021), 100075. pmid: 34027512.
- [10] N. K. Shrestha et al.: Necessity of COVID-19 vaccination in previously infected individuals. medRxiv (2021). doi: 10.1101/2021.06.01.21258176.
- [11] S. S. Nielsen et al.: SARS-CoV-2 elicits robust adaptive immune responses regardless of disease severity. EBioMedicine 68 (2021), 103410. pmid: 34098342.
- [12] A. Grifoni et al.: Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 181 (2020), 1489–1501.e15. pmid: 32473127.
- [13] N. Le Bert et al.: SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature 584 (2020), 457–462. pmid: 32668444.
- [14] S. Cao et al.: Post-lockdown SARS-CoV-2 nucleic acid screening in nearly ten million resi- dents of Wuhan, China. Nat. Commun. 11 (2020), 5917. pmid: 33219229.
- [15] R. Wölfel et al.: Virological assessment of hospitalized patients with COVID-2019. Nature 581 (2020), 465–469. pmid: 32235945.
- [16] K. Basile et al.: Cell-based culture of SARS-CoV-2 informs infectivity and safe de-isolation assessments during COVID-19. Clin. Infect. Dis. (2020). pmid: 33098412.
- [17] Anonymous: Covid: Secret filming exposes contamination risk at test results lab. 2021. url: https://www.bbc.com/news/uk-56556806.
- [18] K. G. Andersen et al.: The proximal origin of SARS-CoV-2. Nat. Med. 26 (2020), 450–452. doi: 10.1038/s41591-020-0820-9.
- [19] B. Sørensen et al.: Biovacc-19: A Candidate Vaccine for Covid-19 (SARS-CoV-2) Developed from Analysis of its General Method of Action for Infectivity. QRB Discovery 1 (2020). doi: 10.1017/qrd.2020.8.
- [20] B. Sørensen et al.: The evidence which suggests that this is no naturally evolved virus. Preprint (2020). url: https : / / www . minervanett . no / files / 2020 / 07 / 13 / TheEvidenceNoNaturalEvol.pdf.
- [21] L. Yan et al.: Unusual Features of the SARS-CoV-2 Genome Suggesting Sophisticated Labora- tory Modification Rather Than Natural Evolution and Delineation of Its Probable Synthetic Route. Preprint (2020). doi: 10.5281/zenodo.4028829.
- [22] L. Yan et al.: SARS-CoV-2 Is an Unrestricted Bioweapon: A Truth Revealed through Uncov- ering a Large-Scale, Organized Scientific Fraud. Preprint (2020). doi: 10.5281/zenodo. 4073131.
- [23] S. Yang and R. E. Rothman: PCR-based diagnostics for infectious diseases: uses, limitations, and future applications in acute-care settings. Lancet Infect. Dis. 4 (2004), 337–48. pmid: 15172342.
- [24] V. M. Corman et al.: Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill. 25 (2020). pmid: 31992387.
- [25] Anonymous: Corman-Drosten review report. 2020. url: https://cormandrostenreview. com/.
- [26] R. Jaafar et al.: Correlation Between 3790 Quantitative Polymerase Chain Reaction-Positives Samples and Positive Cell Cultures, Including 1941 Severe Acute Respiratory Syndrome Coronavirus 2 Isolates. Clin. Infect. Dis. 72 (2020), e921. pmid: 32986798.
- [27] F. M. Liotti et al.: Assessment of SARS-CoV-2 RNA Test Results Among Patients Who Re- covered From COVID-19 With Prior Negative Results. JAMA internal medicine 181 (2020), 702–704. pmid: 33180119.
- [28] J. Bullard et al.: Predicting Infectious Severe Acute Respiratory Syndrome Coronavirus 2 From Diagnostic Samples. Clin. Infect. Dis. 71 (2020), 2663–2666. pmid: 32442256.
- [29] Anonymous: FDA briefing document: Pfizer-BioNTech COVID-19 Vaccine. 2020. url: https: //www.fda.gov/media/144245/download.
- [30] Anonymous: Assessment report/Comirnaty. 2021. url: https://www.ema.europa.eu/ en/documents/assessment-report/comirnaty-epar-public-assessment-report_ en.pdf.
- [31] R. W. Frenck et al.: Safety, Immunogenicity, and Efficacy of the BNT162b2 Covid-19 Vaccine in Adolescents. N. Engl. J. Med. (2021). pmid: 34043894.
- [32] R. A. Campbell et al.: Comparison of the coagulopathies associated with COVID-19 and sep- sis. Research and practice in thrombosis and haemostasis 5 (2021), e12525. pmid: 34027292.
- [33] G. H. Frydman et al.: The Potential Role of Coagulation Factor Xa in the Pathophysiology of COVID-19: A Role for Anticoagulants as Multimodal Therapeutic Agents. TH open : com- panion journal to thrombosis and haemostasis 4 (2020), e288–e299. pmid: 33043235.
- [34] Anonymous: SARS-CoV-2 mRNA Vaccine (BNT162, PF-07302048) 2.6.4 [Summary statement of the pharmacokinetic study] (Japanese). 2020. url: https://www.pmda.go.jp/drugs/ 2021/P20210212001/672212000_30300AMX00231_I100_1.pdf.
- [35] I. C. Kourtis et al.: Peripherally administered nanoparticles target monocytic myeloid cells, secondary lymphoid organs and tumors in mice. PLoS One 8 (2013), e61646. pmid: 23626707.
- [36] C. Ye et al.: Co-delivery of GOLPH3 siRNA and gefitinib by cationic lipid-PLGA nanoparticles improves EGFR-targeted therapy for glioma. J. Mol. Med. Berl. 97 (2019), 1575–1588. pmid: 31673738.
- [37] R. Dal Magro et al.: ApoE-modified solid lipid nanoparticles: A feasible strategy to cross the blood-brain barrier. J. Control. Release 249 (2017), 103–110. pmid: 28153761.
- [38] R. B. Brown: Public health lessons learned from biases in coronavirus mortality overestima- tion. Disaster Med. Public Health Prep. (2020), 1–24. pmid: 32782048.
- [39] V. Furer et al.: Herpes zoster following BNT162b2 mRNA Covid-19 vaccination in patients with autoimmune inflammatory rheumatic diseases: a case series. Rheumatology (2021). pmid: 33848321.
- [40] S. M. C. Tirado and K.-J. Yoon: Antibody-dependent enhancement of virus infection and disease. Viral immunology 16 (2003), 69–86. pmid: 12725690.
- [41] C.-T. Tseng et al.: Immunization with SARS coronavirus vaccines leads to pulmonary im- munopathology on challenge with the SARS virus. PLoS One 7 (2012), e35421. pmid: 22536382.
- [42] F. Negro: Is antibody-dependent enhancement playing a role in COVID-19 pathogenesis? Swiss Med. Wkly. 150 (2020), w20249. pmid: 32298458.
- [43] J. A. Tetro: Is COVID-19 receiving ADE from other coronaviruses? Microbes and infection 22 (2020), 72–73. pmid: 32092539.
- [44] Y. Honda-Okubo et al.: Severe acute respiratory syndrome-associated coronavirus vaccines formulated with delta inulin adjuvants provide enhanced protection while ameliorating lung eosinophilic immunopathology. J. Virol. 89 (2015), 2995–3007. pmid: 25520500.
Featured image is from Children’s Health Defense
By Dr. Michael Palmer, Dr. Sucharit Bhakdi, and Dr. Stefan Hockertz
Join: 👉 https://t.me/acnewspatriots
The opinions expressed by contributors and/or content partners are their own and do not necessarily reflect the views of AC.NEWS
Disclaimer: This article may contain statements that reflect the opinion of the author. The contents of this article are of sole responsibility of the author(s). AC.News will not be responsible for any inaccurate or incorrect statement in this article www.ac.news websites contain copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available to our readers under the provisions of “fair use” in an effort to advance a better understanding of political, health, economic and social issues. The material on this site is distributed without profit to those who have expressed a prior interest in receiving it for research and educational purposes. If you wish to use copyrighted material for purposes other than “fair use” you must request permission from the copyright owner. Reprinting this article: Non-commercial use OK. If you wish to use copyrighted material for purposes other than “fair use” you must request permission from the copyright owner.
Disclaimer: The information and opinions shared are for informational purposes only including, but not limited to, text, graphics, images and other material are not intended as medical advice or instruction. Nothing mentioned is intended to be a substitute for professional medical advice, diagnosis or treatment.
























![Tucker Carlson Released an ALARMING Message … [Published Yesterday]](https://ac.news/wp-content/uploads/2024/04/download-3-120x86.jpg)





















Discussion about this post