A well known antimicrobial, Nitric Oxide, has been found to rapidly reduce SARS-CoV-2 viral load, knocking it down by 95% within 24 hours, and 99% within 72 hours, according to a recent study by researchers funded by England’s NHS foundation trust and SaNOtize Research & Development Corporation – a Canadian biotech company currently conducting Phase II trials of a nitric oxide nasal spray.
A group of 80 adults (18-70 years) with confirmed (Alpha strain) Covid-19 infections were divided into two groups, with half receiving nitric oxide nasal spray (NONS) that were self-administered 5-6 times daily for 9 days.
The goal of the nasal spray is to kill the virus present in the upper airways – preventing it from incubating and making its way to the lungs.
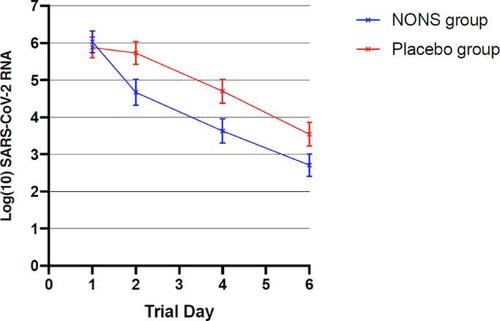
The study found that mean viral load was significantly lower in the NONS group by a factor of 16.2, in what the study’s authors described as an “accelerated decrease,” while nearly half of those who completed a post-study questionnaire reported feeling better vs. 8% of the placebo group.
Mean SARS-CoV-2 RNA concentration was lower on NONS by a factor of 16.2 at days 2 and 4. A rapid reduction (95%) in the SARS-CoV-2 viral load was observed within 24 hours, with a 99% reduction observed within 72 hours with NONS treatments. -Clinical efficacy of nitric oxide nasal spray (NONS) for the treatment of mild COVID-19 infection.
What’s more, there were no serious adverse reactions from the nasal spray.
“Treatment with NONS in this trial was found to be effective and safe in reducing the viral load in patients with mild, symptomatic COVID-19 infection,” reads the study. “Patients in the NONS treatment arm demonstrated viral loads, as determined from PCR testing of nose and throat swab sampling, that were lower at days 2 and 4 by a factor of 16.2 than those on placebo, and symptom resolution was also found to be faster on NONS treatment than on placebo in this study.”
Lower SARS-CoV-2 RNA loads in patients with NONS may be beneficial in the prevention of SARS-CoV-2 transmission. It has been described that higher viral loads in patients with SARS-CoV-2 earlier than SARS-CoV may have contributed to greater difficulties in reducing the onward transmission. Furthermore, it has been observed that the risk of symptomatic COVID-19 was associated with the SARS-CoV-2 RNA levels of contacts and incubation time was shortened in a dose-dependent manner.
Accelerated SARS-CoV-2 clearance with NONS may reduce symptom duration, decrease infectivity period, reduce hospital admissions, and lower disease severity. Consequently, this study could be used as supporting evidence for emergency use of NONS for patients with mild COVID-19 infection.
According to SaNOtize, researchers from Utah State University were able to kill 99.9% of SARS-CoV-2 in a petri dish within two minutes.
The company, whose board includes Prof. Ferid Murad of Stanford University – who won the Nobel Prize in 1998 for discovering the properties of nitric oxide, signed an agreement with Indian biotech Glenmark earlier this month to manufacture, market and distribute NONS throughout Asia, including Singapore, Malaysia Hong Kong, Taiwan, Nepal, Brunei, Cambodia, Laos, Myanmar, Sri Lanka, Timor-Leste and Vietnam.
According to SaNOtize Chief Science Officer, Dr. Chris Miller, the nasal spray is a ‘post-exposure’ prevention akin to hand sanitizer.
“If you are outside, around people, and could be infected, you could use the spray and reduce the number of viruses in the nose, before it is becoming a full-blown infection. We have shown that even when people have a very high load of virus, the spray can significantly reduce the viral load,” Miller said in May.
by Tyler Durden
Join: 👉 https://t.me/acnewspatriots
The opinions expressed by contributors and/or content partners are their own and do not necessarily reflect the views of AC.NEWS
Disclaimer: This article may contain statements that reflect the opinion of the author. The contents of this article are of sole responsibility of the author(s). AC.News will not be responsible for any inaccurate or incorrect statement in this article www.ac.news websites contain copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available to our readers under the provisions of “fair use” in an effort to advance a better understanding of political, health, economic and social issues. The material on this site is distributed without profit to those who have expressed a prior interest in receiving it for research and educational purposes. If you wish to use copyrighted material for purposes other than “fair use” you must request permission from the copyright owner. Reprinting this article: Non-commercial use OK. If you wish to use copyrighted material for purposes other than “fair use” you must request permission from the copyright owner.
Disclaimer: The information and opinions shared are for informational purposes only including, but not limited to, text, graphics, images and other material are not intended as medical advice or instruction. Nothing mentioned is intended to be a substitute for professional medical advice, diagnosis or treatment.


















![Tucker Carlson Released an ALARMING Message … [Published Yesterday]](https://ac.news/wp-content/uploads/2024/04/download-3-120x86.jpg)






![BENJAMIN FULFORD GEOPOLITICAL UPDATE 4⧸1⧸24…AUDIO READING [mirrored]](https://ac.news/wp-content/uploads/2024/04/download-16-120x86.jpeg)



















Discussion about this post